

Development of an Activated Carbon Fiber Cloth Adsorption/Regeneration System to Recover and Reuse Toxic Volatile Organic Compounds |
SYSTEM EVALUATION EXPERIMENTS -- Continued
Mass Transfer Model
The thermodynamic model coupled with the Wagner equation, used to determine the saturation TVOC concentration, provides a method to estimate outlet concentration, removal efficiency and refrigerant requirement . However, this model assumes thermodynamic equilibrium and no mass resistance. Assuming no mass transfer resistance may yield condenser designs in which equilibrium between the TVOC vapor and the corresponding condensate film is not achieved. Outlet concentrations will be higher in undersized non-equilibrium condensers. The thermodynamic model is not dependent on the physical shape and size of the condenser, which is an important design consideration.
Two transfer mechanisms influence the selection of condenser size based on desired TVOC removal: 1) mass transfer surface area required to achieve the desired mass removal (Chilton and Colburn, 1934) and 2) heat transfer surface area required to achieve the desired condensation temperature, thus mass removal (Colburn and Hougen, 1934). These mechanisms should be coupled together with momentum transport and the fundamental thermodynamics of the system. Condenser size based on desired mass removal is determined from the surface area required to transport the desired mass to the surface (mass transfer) and the surface area required to achieve the desired condenser temperature (heat transfer and thermodynamics).
Mass transfer coupled with momentum transport and thermodynamics can be evaluated independent of heat transfer if the condenser temperature profile is assumed or if an average condenser temperature is determined directly from the thermodynamic model described above. Determining the condenser temperature from thermodynamic equilibrium assumes that heat transfer resistance is negligible. The effect of heat transfer resistance is to increase the temperature (thus outlet TVOC concentration) of the challenge gas stream predicted from the thermodynamic model.
Mass transfer condensation is modeled for an indirect contact condenser in which the refrigerant gas stream is not mixed with the challenge gas stream. The challenge gas stream is modeled as forced convection of a vapor in a non-condensable gas condensing into a liquid film. Essentially all condensation in a shell-and-tube heat exchanger is filmwise as opposed to dropwise (Wilbur, 1985). Mass transfer occurs from the bulk gas flow of the challenge gas stream to the condensate film. A gas film is established between the bulk flow and condensate layer which resists mass transfer as discussed in section 3.
The mass transfer driving force results from a concentration gradient in the gas film (Figure 4.31). The temperature at the gas/condensate interface is lower than in the bulk challenge gas stream. The lower temperature results in a lower saturation concentration at the interface than in the bulk flow if the vapor is at the saturation point. This concentration gradient induces mass transport to the condensate surface where condensation occurs (Chilton and Coulburn, 1934).
Figure 4.31 Condenser element used for mass and heat transfer models.
The mass transfer model incorporates the thermodynamic model discussed above with the radial mass transport of the vapor to the condensate surface. The following assumptions are made in addition to the assumptions made in the thermodynamic model development:
1) laminar challenge gas flow (Re < 2400)
2) adiabatic conditions
3) temperature at the condensate film is the thermodynamic equilibrium temperature T2 from the thermodynamic model if this module is used
4) condensation surface is modeled using the flat plate assumption (Rosenow et al., 1985)
5) gas and liquid properties are dependent on temperature, composition and concentration only
6) complete vaporization of the LN2
7) temperature and concentration gradients are log-mean (Wilbur, 1985)
8) vapor equilibrium concentration exists at the gas/condensate interface (Kern, 1950) (found from the Wagner equation)
9) condensation occurs at the condensate surface only (Rosenow et al., 1985)
10) no condensate surface waves; waviness increases mass transfer (Siddique et al., 1994)
11) uniform condensate film thickness (wash-out is equal to mass condensed)
12) driving mass transfer force is from the concentration gradient between bulk challenge gas and condensate film (Chilton and Coulburn, 1934)
13) the TVOC mass entering the gas film boundary layer is condensed (Kern, 1950)
To determine axial concentration of a vapor in the condenser, radial mass transfer is determined from a mass transfer coefficient. The mass transfer coefficient, Kg, is developed from the Chilton and Coulburn analogy between and mass transfer (1934):
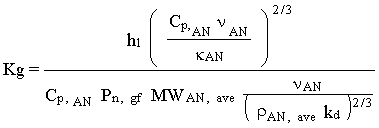 (4.15)
(4.15)
where,
h1 = heat transfer coefficient for laminar flow with the entrance length correction
Cp,AN = specific heat of the VOC-carrier gas challenge gas stream
< AN = viscosity of the VOC-carrier gas challenge gas stream
kAN = thermal conductivity of the VOC-carrier gas challenge gas stream
Pn,gf = log-mean partial pressure of carrier gas in the mass transfer boundary layer
MWAN,ave = average molecular weight of the challenge gas stream
rAN,ave = average density of the challenge gas stream
kd = diffusion coefficient of TVOC in carrier gas using the Gilliand equation
This equation is shown to conservatively hold for flow inside tubes, flow across a single tube and flow along plane surfaces (Kern, 1950). The appropriate value for h1 is substituted into the equation depending on the flow conditions. Determination of the convective heat transfer coefficient involves the Nusselt analysis. The Nusselt number is given as (Incropera and DeWitt, 1990):
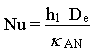
Seider and Tate (1936) showed that for laminar flow inside circular tubes with a constant surface temperature, the Nusselt number is given as:
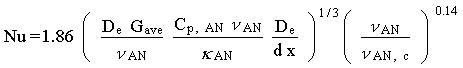
where Tc is constant and:
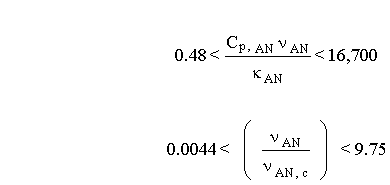
De = effective heat transfer shell-and-tube diameter,
Gave = average radial mass flow rate of TVOC to the condensate surface,
dx = condenser element length,
< AN,c = VOC-carrier gas viscosity at the condensate surface and
TC = temperature of condensate film.
Equation 4.18 includes the combined entry length correction. This correction is used for condensers in which the challenge gas stream does not have fully developed velocity and thermal profiles at the entrance. Whitaker (1972) recommends this equation for:
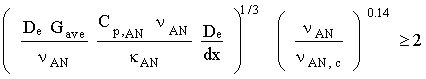
Otherwise, fully developed flow exists in the majority of the shell in which the challenge stream transverses, and an approximation for the Nusselt number is (Incropera and DeWitt, 1990):
Nu = 3.66 (4.19)
The mass transfer model in Appendix D is written for the entrance length correction. Equating (4.16) and (4.17) and solving for h1 results in the convective heat transfer coefficient:
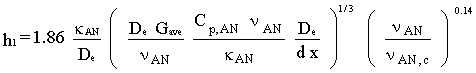
The diffusion coefficient (kd) in eq. (4.2) is determined from the empirical Gilliand equation for the diffusivity of one gas through another (Gilliand, 1934):
where,
TA1 = temperature of bulk challenge gas at the element entrance
TA2 = temperature of bulk challenge gas at the element exit
P = ambient pressure
nN = viscosity of the carrier gas in the challenge gas stream
nA = viscosity of the TVOC in the challenge gas stream
MWN = molecular weight of carrier gas in the challenge gas stream
MWA = molecular weight of the TVOC in the challenge gas stream
The amount of TVOC condensed for each element can be determined from a mass balance of the TVOC entering the gas film. Based on the assumption that all of the mass of TVOC entering the gas is condensed, the mass transfer coefficient can be used to determine the mass condensed (Kern, 1950):
MLA = Kg dPlm,vapor SA MWvapor (4.22)
where,
MLA = mass of acetone condensed
Kg = mass transfer coefficient
dPlm,vapor = log-mean vapor partial pressure difference between the bulk VOC-carrier gas flow and condensate film
SA = condensation surface area
MWvapor = molecular weight of acetone
The required condensation surface area can be determined from eq. (4.14).
The model in Appendix D was written using acetone as the adsorbate and N2 as the carrier gas. Different compounds may be evaluated by substituting the appropriate material property equations and constants into the model. The list of variables found in Appendix B details the constants used in determining material properties. The fundamental mass transfer coefficient, Kg, diffusion coefficient, kd, heat transfer coefficient, h1, and thermodynamic equations are applicable as long as the above assumptions are adequate for evaluation purposes. Also, modeled specific heats, conductivities and visocities were verified with experimental data presented in literature (Carmichael, 1996).
The mass transfer model provides a method of determining condenser mass removal performance, refrigerant flow rate and required surface area based on mass transfer. Typical inputs to the model include inlet gas temperature, inlet gas flow rate, inlet TVOC concentration and either outlet concentration or refrigerant flow rate. The refrigerant flow rate will establish the condenser temperature profile, and thus from the Wagner equation and mass transfer, the axial concentration profile. Therefore if the refrigerant flow rate is defined then the condenser axial concentration is found or vice versa. Defining an outlet concentration will fix the refrigerant flow rate required and the axial concentration profile. Another option is to circumvent the thermodynamic module and input the temperature profile directly.
If the refrigerant flow rate is used, the model determines a condenser temperature based on thermodynamic equilibrium. The modeled bulk gas temperature in the condenser will be lower than the actual temperature due to heat transfer resistance that is not considered in this model. Using the thermodynamic model will under predict the axial concentration at any point in the condenser. Higher refrigerant flow rates will be required to counter heat transfer resistance and achieve the desired condenser temperature (thus outlet concentration).
The outlet mole fraction was determined as a function of condenser surface area for a gas stream with the following characteristics:
Inlet acetone mole fraction = 0.05 to 0.8
Inlet gas temperature = 294 K
Inlet challenge gas flow rate = 1 actual lpm
Temperature of the condensate = 195 K
Temperature of outlet gas = 200 K
Thus, the temperature profile was directly input in lieu of allowing the thermodynamic module to determine the condenser temperature. This was done to determine the refrigerant flow rate required to achieve these condenser conditions. The average condenser temperature is assumed log-mean (Kern, 1950).
The minimum TVOC outlet concentration is predicted at approximately 1000 cm2 for an inlet acetone mole fraction of 0.25 (Figure 4.32). The LN2 flow rate was found to be 10.1 g/sec to achieve the inputted condenser temperature profile. Results show that outlet TVOC concentration cannot be changed appreciably at large condenser surface areas. This is a result of the bulk acetone vapor concentration approaching the saturation equilibrium concentration at the condensate temperature.
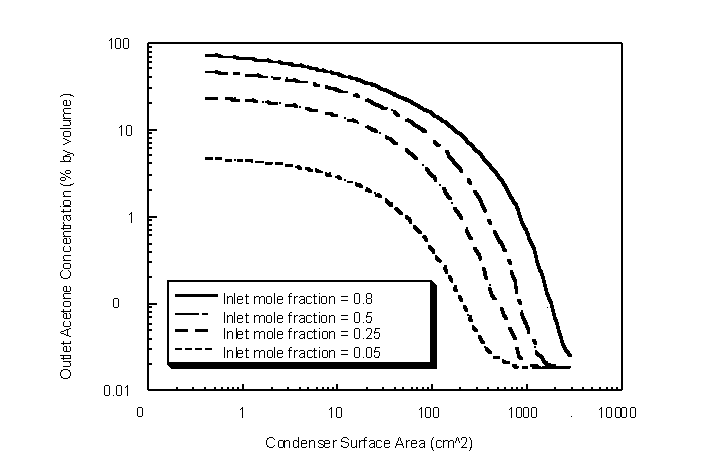
Figure 4.32 Modeled outlet acetone volume fractions for various condenser lengths. Inlet temperature = 294 K, outlet temperature = 200 K, condensate film temperature = 195 K and challenge gas stream flow rate = 1 lpm.
A modeled plot of acetone mole fraction versus condenser surface area for specific process conditions can be made for design purposes. Knowing the desired outlet concentration, the required mass transfer condensation surface area and refrigerant flow rate can be determined to achieve the desired outlet concentration (removal efficiency).
Modeling provides an analysis of condensers that can be used in determining an optimum design. The thermodynamic model provides insight into removal efficiencies, outlet concentrations and required refrigerant flow rates given process conditions. Incorporating mass transfer with the thermodynamic model provides a method for determining the condensation surface area required to achieve the desired outlet concentration
Cryogenic Condensation Modeled and Experimental Results
The objective of developing the bench-scale cryogenic condensation system is to evaluate the feasibility of condensing TVOC vapors using LN2 as a refrigerant. Experimental results are presented in this section to evaluate the performance of the cryogenic condensation system. The condenser outlet TVOC concentration is experimentally determined for various flow rates, inlet concentrations and condenser temperatures. Also, initial and optimized test results are presented for the integrated ACFC adsorption/cryogenic condensation system. Model results from the thermodynamic and mass transfer models already presented in this report are also presented.
CO2 Refrigerant Results
A preliminary TVOC condensation test was conducted to evaluate the outlet acetone vapor concentration using an alternative refrigerant. Solid carbon dioxide (CO2, dry ice) was
chosen because of its low temperatures and ease of handling and delivery. A flow rate of 3 actual lpm of house air was passed through the center of a 61 cm length H 7.6 cm diameter hollow cylinder packed with dry ice. The total flow rate of refrigerant (house air and sublimed CO2) was 5.5 actual lpm. This flow was passed through the inside tube of the condenser. The condenser was cooled to 264 " 1 K without the vapor laden gas stream passed through the shell. A 500 sccm gas flow of 85.8 " 2.0% N2 and 14.2 " 2.0% acetone was then introduced counter currently into the shell. The temperature measured at the outlet of the challenge gas stream was 273 " 5 K. The outlet concentration of acetone in gaseous N2 was monitored using syringe samples drawn at the condenser outlet and injected into the GC/MS (Figure 4.33). After approximately 11 min, the average outlet concentration was 8.1 " 0.6% by volume. The maximum theoretical concentration using the Wagner Equation at 273 K is 9.1% (Figure 3.3). The mean mass removal efficiency was 45.6% with CO2-air refrigerant.
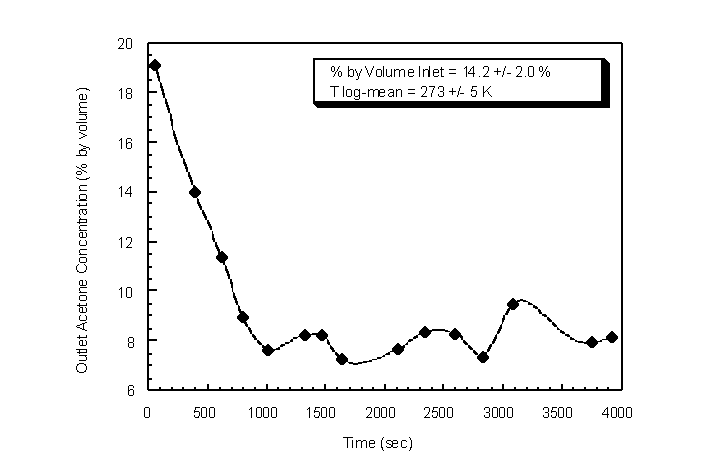
Figure 4.33 Condenser outlet acetone concentration using CO2 and compressed air as the tube refrigerant. Flow rate = 0.5 actual lpm, condenser shell pressure = 745 mm Hg and ambient temperature = 294 K
.
The outlet concentration was near the acetone equilibrium saturation concentration at the temperature of the outlet challenge gas stream. This demonstrates that using air passed over solid CO2 does not achieve temperatures low enough to condense large quantities of acetone unless the inlet acetone gas concentration is above the saturation concentration at ambient temperature. The high outlet acetone concentrations and low removal efficiency excludes this type of refrigerant from many applications. If the concentration of acetone is below the saturation concentration at 273 K then little or no acetone will be condensed.
Condenser Temperature Characteristics
The use of LN2 as a refrigerant provides the cooling capacity to achieve temperatures near the freezing points of many organic compounds (Figure 3.3). At these temperatures, TVOC saturation concentrations are much lower than for the saturation concentrations at temperature ranges for refrigerants such as water, dry ice or ethylene glycol/water mixtures.
Another advantage for the use of LN2 over other refrigerants is that the LN2 undergoes a phase change from a liquid to a gas. The latent heat of vaporization and gaseous sensible heat are utilized for cooling the TVOC and carrier gas. Other refrigerants typically do not undergo a phase change and therefore only sensible heat is utilized for cooling capacity.
An initial test was conducted on the shell-and-tube condenser to determine the time to reach the set-point temperature and to determine the temperature frequency and amplitude of the temperature control system. UHP nitrogen was sent through the shell side at 0.995 slpm in lieu of the vapor laden gas stream. LN2 was then delivered through the tube at a liquid mass flow rate of 5.9 g/min. The pressurized dewar was set at 20 pounds per square inch. The temperature control system was set at 185 K. The temperature of the inner tube wall was measured as a function of time (Figure 4.34).
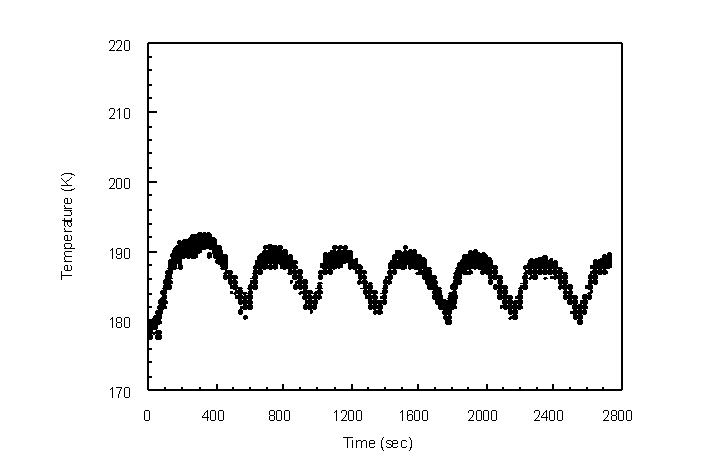
Figure 4.34 Temperature cycling of the condenser using the temperature control system at a set-point of 185 K.
The set-point temperature was reached after 5490 sec at which time the solenoid valve began cycling (Figure 4.35). The valve remained open for an average of 300 sec and closed for an average of 92 sec per cycle. The temperature was controlled within +6 K and -5 K of the set-point temperature.
Figure 4.35 Condenser cool-down to set point cycling
Modeled and Experimental Refrigerant Requirements
Theoretical and experimental LN2 refrigerant requirements were evaluated as a function of inlet acetone concentrations (Figure 4.36). The theoretical mass of LN2 per unit mass of acetone condensed was determined from the thermodynamic model developed in section 4. Experimental LN2 mass was determined from the outlet gas flow rate of N2.
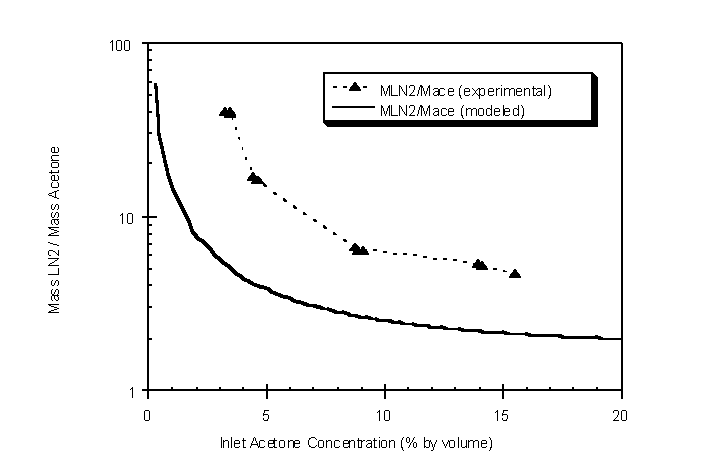
Figure 4.36 Modeled and experimental LN2 requirements as a function of inlet acetone concentration
With the desired inlet acetone concentration flowing through the shell, the condenser was cooled to steady-state temperature cycling. The temperature and flow rate of the N2 gas were measured at the condenser outlet and corrected to 298 K and 1 atm. An average flow rate was determined for one complete cycle in which the solenoid valve was opened and closed. The mass of LN2 was calculated for each complete temperature cycle using the volume expansion ratio for LN2 to gaseous N2 (1:696) at standard conditions (298 K and 1 atm). The mass of acetone condensed was determined by measuring inlet and outlet acetone gas concentrations, flow rate and time for each temperature cycle. The mass of condensed acetone is thereby determined from:
mass acetone condensed ~ (Cin - Cout) H Qin H t (4.21)
where,
Cin = inlet acetone concentration
Cout = outlet acetone concentration
Qin = flow rate of the inlet gas stream
t = one complete temperature cycle
Deviations of the experimental data from the modeled data are most likely due to heat entering the condenser from the outside. The model assumes adiabatic conditions in which heat is not transferred into or out of the system except from the liquid condensate washing out. The condenser system insulation does not completely eliminate all heat transfer.
Modeled and experimental results show that at low inlet concentrations more LN2 is required to condense a unit mass of acetone (Figure 4.36). For instance, at an inlet concentration of 2.0% by volume acetone, 10 kg of LN2 is theoretically required to condense 1 kg of acetone. However, at an inlet concentration of 20% by volume acetone, only 2 kg of LN2 is required to condense 1 kg of acetone. Experimental results show the same general trend. This supports the earlier statement that higher concentration vapor streams result in more efficient use of LN2. High outlet TVOC concentrations can generally be obtained from desorption of activated carbon after breakthrough of the TVOC has been achieved. The use of a carbon adsorber upstream of the condenser facilitates the more efficient use of LN2.
Condenser Performance Evaluation
Condenser outlet acetone gas concentrations were measured for various different possible operating conditions to gain better insight into condenser performance. Modeled results are presented along with experimental results where applicable. Condenser temperature data are presented with the performance curves due to the dependence of the outlet concentration on temperature (Wagner equation).
Condenser outlet acetone gas concentration was measured for highly variable inlet acetone concentrations. Ambient temperature and pressure were 294 K and 743 mm Hg, respectively, with a 0.5 actual lpm challenge gas flow rate passing through the condenser shell. The condenser was cooled to steady-state temperature cycling prior to the introduction of the challenge gas stream (Figure 4.37). LN2 was not passed through the outside cooling coil for this experiment to compare results with tests that utilize the coil. Inlet acetone gas concentrations were varied between 2% and 20% by volume acetone to determine the condenser outlet acetone concentrations (Figure 4.38).
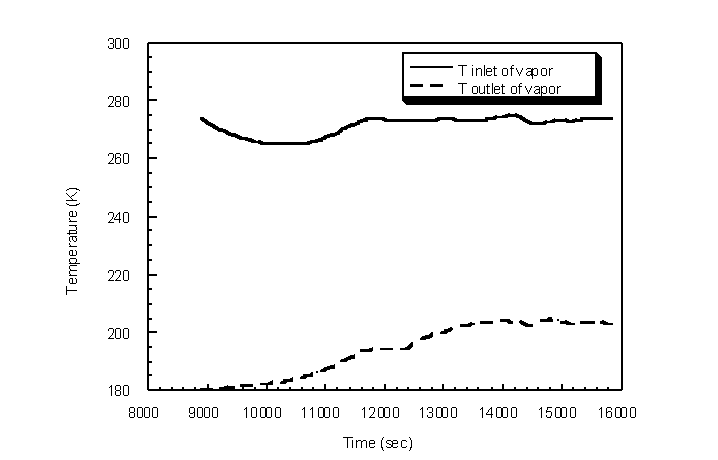
Figure 4.37 Condenser temperature profile for variable inlet acetone concentrations
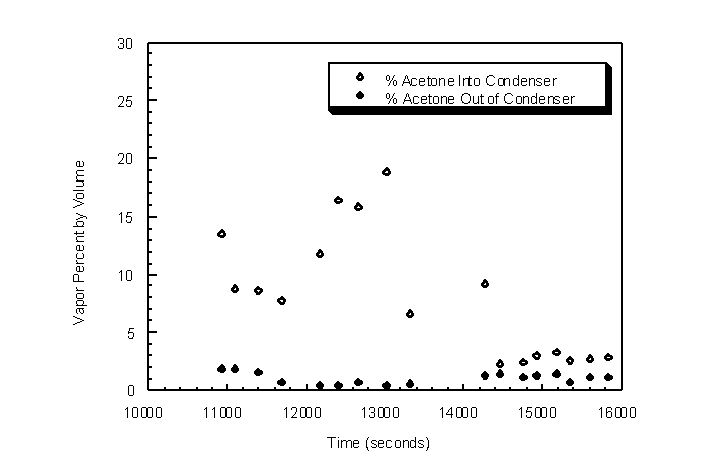
Figure 4.38 Condenser inlet and outlet concentrations for variable inlet acetone concentrations
The outlet acetone gas concentration remained fairly constant for abrupt changes in inlet concentration (Figure 4.38). The outlet acetone gas concentrations ranged between 0.4% and 1.9% by volume corresponding to concentrations between 0.1% and 2.0% predicted by the Wagner equation. The relatively high outlet acetone concentrations are due to the relatively high condenser temperature at the vapor inlet. To create a more uniform condenser temperature profile, 3/8 inch diameter spherical brass packing was packed in the condenser tube. The packing provides mass that resists temperature change, and the brass material conducts heat from the relatively warmer vapor inlet to the cooler vapor outlet. A more uniform temperature profile was achieved as will be shown later with the packed heat exchanger. The limiting condenser temperature is the freezing temperature of the TVOC or H2O if H2O exists in the gas stream. Lower log-mean condenser temperatures can be achieved by bringing the inlet and outlet condenser temperatures closer together. In effect, the lower temperature is raised and the higher temperature is lowered. Because the lower temperature is raised, higher mass flow rates of LN2 can be utilized to decrease the lower condenser temperature back near the freezing point of the VOC, while the higher temperature is decreased overall. The TVOC therefore has a higher residence time in a lower overall condenser temperature profile, thereby promoting further condensation of VOC.
Another test was conducted with conditions similar to that outlined in the previous test. Ambient temperature was 294 K and ambient pressure was 748 mm Hg. The challenge gas inlet flow rate was 0.5 actual lpm. Step inlet acetone gas concentrations ranging from 9.5% to 3.0% by volume were passed through the condenser shell. LN2 was passed through the tube packed with the brass spheres. LN2 was also passed through the outside shell coil. The objective of this experiment was to determine if the temperature profile and outlet acetone concentration could be reduced.
The condenser temperature at the inlet of the vapor was reduced by 15 " 10 K utilizing the brass packing and the cooling coil (Figure 4.39). Correspondingly, the outlet acetone concentrations were reduced from a range between 2.0% and 0.1% by volume down to a range between 0.3% and 0.04% by volume (Figure 4.40). The average outlet acetone gas concentration is 0.19% by volume. The Wagner equation predicts an acetone concentration of 0.20% by volume for a log-mean condenser temperature of 221 K.
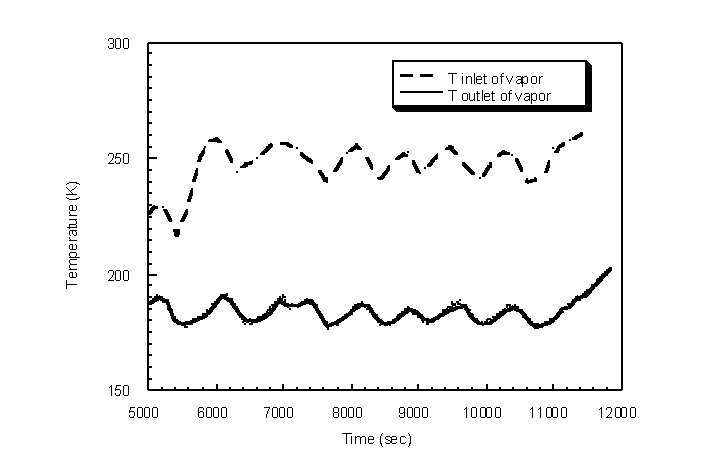
Figure 4.39 Condenser temperature profile for step acetone inlet concentrations
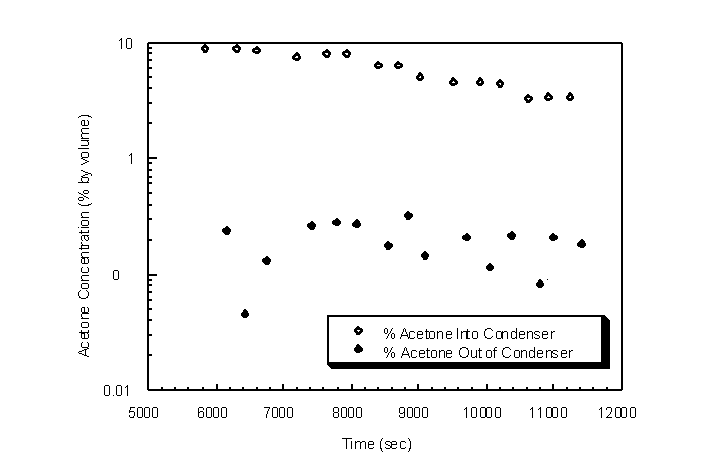
Figure 4.40 Condenser inlet and outlet acetone concentrations for step inlet concentrations
Condenser TVOC outlet concentrations were also measured for varying TVOC inlet concentrations between 0.25% and 18.3% by volume to determine TVOC removal efficiencies (Figure 4.41).
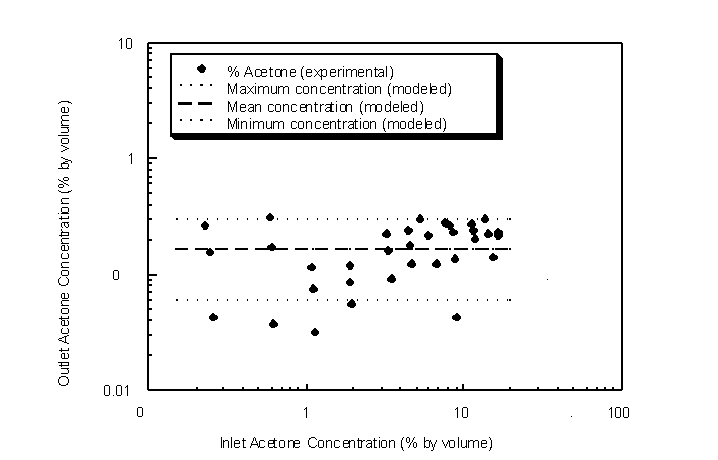
Figure 4.41 Outlet versus inlet acetone concentrations. Modeled maximum, mean and minimum concentrations are the saturation equilibrium concentrations at 225 K, 215 K and 206 K, respectively (experimental condenser temperature range).
The challenge gas flow rate was 0.5 actual lpm and at an ambient temperature of 294 K and ambient pressure of 745 mm Hg. The condenser was pre-cooled to an equilibrium log-mean temperature of 215 " 10 K for each experiment. Over the entire range, outlet acetone concentrations remained fairly constant near the equilibrium vapor concentration of 0.17% at 215 K as predicted by the Wagner equation. Removal efficiencies are therefore higher for higher inlet concentrations. For instance, the removal efficiency found for an acetone inlet concentration of 18.3% was found to be 98.8%. However, the removal efficiency for an inlet acetone concentration of 0.6% was 70.5%.
The inlet and outlet acetone gas concentrations from the above experiment were used to determine removal efficiencies (Figure 4.42). Removal efficiency was also modeled using the thermodynamic model coupled with the Wagner equation. The experimental log-mean condenser temperatures and inlet acetone gas concentrations were input into the model to determine outlet concentrations and refrigerant mass requirements.
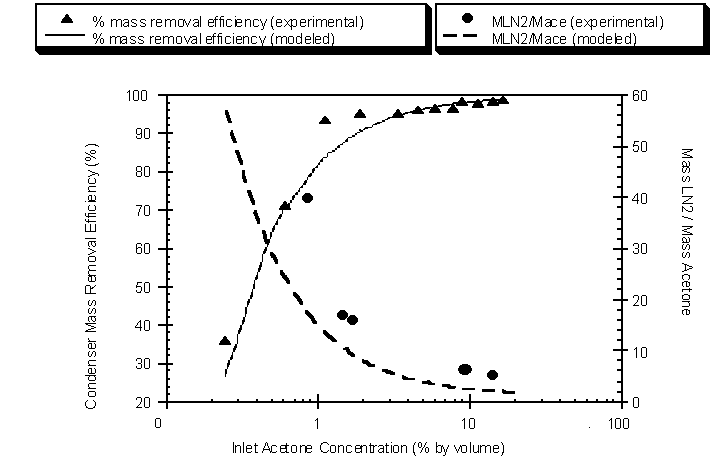
Figure 4.42 Modeled and experimental mass removal efficiencies and refrigerant requirements as a function of the condenser inlet acetone gas concentration
Removal efficiency increases as the inlet concentration of the acetone increases. Also, the mass of LN2 required to condense a unit mass of acetone decreases as inlet acetone gas concentrations increase. Therefore, higher inlet acetone gas concentrations result in higher removal efficiencies and more efficient use of the LN2 refrigerant. Industrial TVOC gas streams typically contain low TVOC concentrations at high flow rates (Ruddy and Carroll, 1993). At low TVOC concentrations, the condenser’s removal efficiency decreases and requires elevated refrigerant requirements. However the use of a carbon adsorber prior to condensation can result in concentrated TVOC levels during desorption. Pre-concentrating the TVOC increases removal efficiency and decreases the amount of LN2 required to condense a unit mass of VOC. As discussed earlier, pre-concentrating the TVOC can also decrease the carrier gas flow sent to the condenser. The decreased flow reduces the condenser sizing requirements by increasing residence time. Overall, carbon adsorption/desorption prior to condensation results in: 1) increased removal efficiency, 2) efficient refrigerant use, and 3) reduction of condensation surface area by 1) increasing the TVOC concentration and 2) reducing the flow rate.
The effect of flow rate on condenser outlet acetone gas concentration was determined for flow rates between 0.5 actual lpm and 3.0 actual lpm. Ambient temperature and pressure were 295 K and 746 mm Hg, respectively. An inlet acetone concentration between 9.1% and 9.5% by volume was passed through the shell of the condenser. The average temperature at each flow rate was determined from thermocouple measurements for modeling purposes (Figure 4.43).
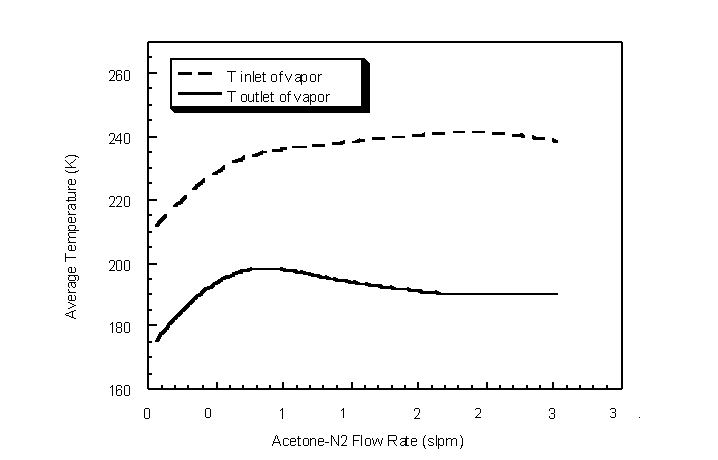
Figure 4.43 Average condenser temperature profile for each flow rate in which inlet and outlet acetone gas concentrations were sampled
The outlet gas concentration remained fairly constant over the flow rate range examined (Figure 4.44). The lower modeled and experimental acetone outlet concentrations for flow rates under 0.5 actual lpm are a result of lower log-mean condenser temperatures. The lower temperatures result in lower saturation concentrations (Wagner equation). The relatively constant outlet acetone concentrations show that the bench-scale cryogenic condenser can be operated at variable flow rates (25 < Re < 1300) without affecting condensation performance. In addition, the constant outlet concentrations indicate that sufficient condensation surface area exists for the vapor to be in equilibrium with its condensate liquid film.
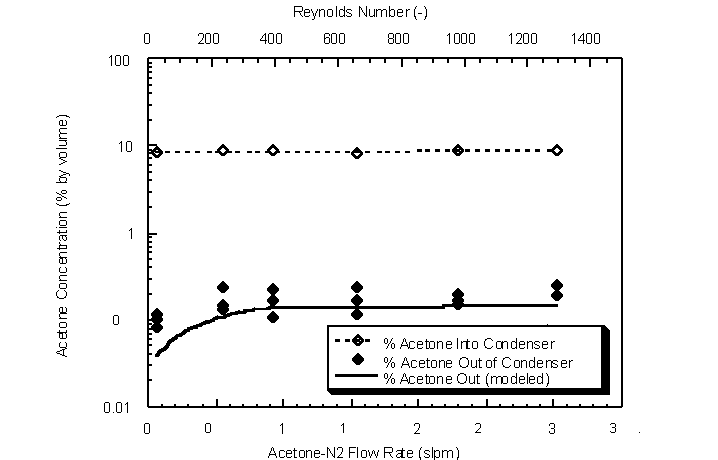
Figure 4.44 Condenser outlet acetone concentrations for various challenge gas flow rates
The condenser was also evaluated for acetone removal performance at various condenser equilibrium temperatures (Figure 4.45). Ambient temperature and pressure were 296 K and 750 mm Hg, respectively. A 10% by volume acetone challenge gas stream at 0.5 actual lpm was sent through the condenser at log-mean temperatures ranging from 209 K to 271 K. The measured
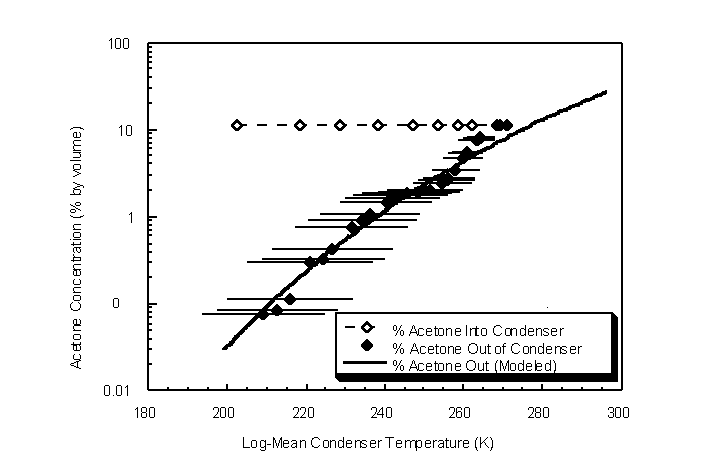
Figure 4.45 Inlet and outlet acetone concentrations for various log-mean condenser temperatures.Error bars are the condenser temperature ranges for each experimental data point
outlet concentrations closely approximated the theoretical equilibrium vapor concentration as predicted by the Wagner equation. This indicates that the acetone bulk vapor concentration reached equilibrium at the liquid-vapor surface on the condenser walls. The model provides a predictive method of determining the outlet concentration given temperature or vice versa.
Outlet concentrations of MEK were also examined for various condenser temperatures (Figure 4.46). Ambient temperature and pressure were 295 K and 746 mm Hg, respectively. A 0.5 actual lpm challenge gas stream was passed through the condenser shell after the desired equilibrium temperature was achieved. Generation of a constant MEK concentration challenge gas stream proved difficult using the dual-bubbler gas generation system. The inlet MEK concentration varied from 8.4% to 18.1% for the duration of the experiment.
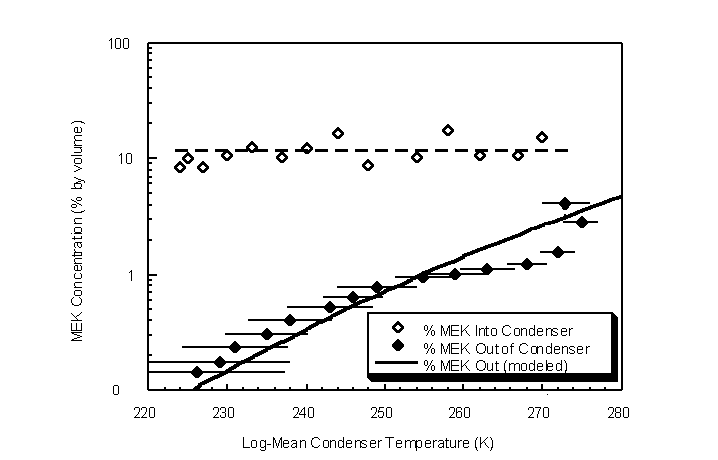
Figure 4.46 Inlet and outlet MEK concentrations for various log-mean condenser temperatures. Error bars are the condenser temperature ranges for each experimental data point
Outlet MEK gas concentrations closely approximated the theoretical modeled saturation curve. This provides evidence that adequate condenser surface area is present for MEK to reach equilibrium with its liquid condensate at each temperature. If sufficient condensation surface area exists for equilibrium between the vapor and liquid to be reached, the modeled saturation curve provides a predictive method to determine the outlet concentration based on a known condenser temperature. Conversely, for design purposes, a known outlet concentration can be used for determining the required condenser temperature.